Life Science 7 Student Text 4th Edition 19b Section Review
Abstract
This report analyzed the potential associations of 7 myocardial fibrosis-related microRNAs with the quality of the collagen network (east.g., the degree of collagen fibril cantankerous-linking or CCL) and the enzyme lysyl oxidase (LOX) responsible for CCL in 28 patients with astringent aortic stenosis (Every bit) of whom 46% had a diagnosis of chronic heart failure (HF). MicroRNA expression was analyzed in myocardial and blood samples. From the studied microRNAs only miR-19b presented a direct correlation (p < 0.05) betwixt serum and myocardium. Compared to controls both myocardial and serum miR-19b were reduced (p < 0.01) in AS patients. In improver, miR-19b was reduced in the myocardium (p < 0.01) and serum (p < 0.05) of patients with HF compared to patients without HF. Myocardial and serum miR-19b were inversely correlated (p < 0.05) with LOX, CCL and LV stiffness in AS patients. In in vitro studies miR-19b inhibition increased (p < 0.05) connective tissue growth factor protein and LOX protein expression in human fibroblasts. In conclusion, decreased miR-19b may be involved in myocardial LOX upwards-regulation and excessive CCL, and consequently increased LV stiffness in AS patients, namely in those with HF. Serum miR-19b can be a biomarker of these alterations of the myocardial collagen network in AS patients, especially in patients with HF.
Introduction
Aortic valve stenosis (Every bit) is the virtually common valve disease in western countries, and its prevalence is age-dependent affecting iii–4% of the population over 75 years formerone. One of the main agin consequences of Every bit is the development of eye failure (HF)2.
Myocardial fibrosis plays a crucial role in the process of myocardial remodelling in patients with Equally, as well equally in the evolution of their clinical manifestationsiii,iv,5, and has a detrimental impact on the improvement in cardiac function, clinical status and long-term survival after aortic valve replacement6,7,viii. Myocardial fibrosis is characterized not only by the increase in the quantity of collagen fibers (i.e., the fraction of myocardium occupied past collagen tissue or CVF) but also by changes in their quality (i.due east., the degree of cross-linking among collagen fibrils or CCL). The degree of CCL determines the stiffness of collagen fibers and their resistance to degradation by matrix metalloproteinases9, and may influence cardiac function and clinical issue. For case, in HF patients it has been shown that an increment in CCL is associated with increased left ventricular (LV) bedchamber stiffness, elevated left-sided filling pressures and the development of diastolic dysfunction10,xi,12, every bit well as with a higher take a chance for hospitalization for HF13. Interestingly, myocardial lysyl oxidase (LOX), the enzyme involved in fibrillary cross-linkingxiv, has been reported to be associated with both CCL and left-sided filling pressures in HF patients11,12.
Several studies take demonstrated that changes in the expression of some microRNAs are associated with myocardial fibrosis (for review see ref. 15). For instance, increased expression of the pro-fibrotic microRNAs miR-21, miR-208a and miR-499-5p16,17,xviii, and decreased expression of the anti-fibrotic microRNAs miR-29, miR-19b, miR-1 and miR-133anineteen,20,21,22,23 have been reported in the myocardium in different cardiac diseases. Interestingly, the circulating levels of some of these microRNAs have been plant to be altered in cardiac patients16,24,25. Nevertheless, the clan of myocardial and circulating microRNAs with both the quantity and the quality of fibrotic tissue in cardiac patients has not been investigated.
Therefore, this study has been designed to analyze the association of the circulating and myocardial expression of the 7 previously mentioned microRNAs with myocardial CVF, CCL and LOX in patients with severe Every bit with and without HF. In addition, in vitro experiments have been performed in human adult fibroblasts with the microRNA of involvement.
Results
Clinical characteristics
Forty-vi pct of AS patients presented HF. The clinical and echocardiographic characteristics of AS patients classified according to the presence or absence of HF are shown in Tables one and 2, respectively. Plasma NT-proBNP was increased (p < 0.01) in Every bit patients with HF compared with those without HF (Table one), fifty-fifty after controlling for age, sexual practice and EF (p ≤ 0.01). The LV stiffness constant (KLV) was higher (p < 0.01) in HF patients compared to non-HF patients (Table 2), even subsequently controlling for age, sexual activity and EF (p ≤ 0.03).
Myocardial fibrosis
As previously published, CVF was increased in Every bit patients compared with command subjects (xviii.41 ± 2.23% vs 1.95 ± 0.07%, p < 0.001), and in all patients the CVF value was above the upper limit of normality in command subjects (95% confidence interval for control subjects: ii.39%)26. In improver, AS patients presented a 3-fold increase (p < 0.001) in soluble collagen (2.00 ± 0.18 μg/mg) and a six-fold increase (p < 0.001) in insoluble collagen (6.15 ± 0.51 μg/mg) as compared with control subjects (0.66 ± 0.17 and 0.96 ± 0.24 μg/mg, respectively). As a consequence, CCL was increased in Equally patients compared with control subjects (3.79 ± 0.40 vs 1.22 ± 0.08, p < 0.001).
It has been previously reported that the expression of LOX was undetectable in myocardial samples from controls, whereas this enzyme was strongly expressed in the myocardium of As patients26. Moreover, LOX expression was straight correlated with CCL (r = 0.703, p < 0.001), and CVF (r = 0.409, p < 0.05), in all Equally patients (Supplementary Figure S1). Moreover, 1000LV was straight correlated with CCL (r = 0.456, p < 0.05), and LOX (r = 0.507, p < 0.01) in all AS patients (Supplementary Figure S2).
Equally patients with HF presented increased (p < 0.01) CVF, CCL and LOX compared to non-HF patients (Table 3), fifty-fifty after controlling for age, sexual practice and EF (p ≤ 0.03).
microRNA expression
From the 7 microRNAs assessed, miR-133a, miR-21 and miR-19b were detected both in myocardial and serum samples from AS patients and command subjects, whereas the expression of miR-29b, miR-1, miR-208a and miR-499-5p was nether the limit of detection in serum samples from As patients and command subjects. Therefore, we focused our study on the former 3 microRNAs.
The expression of miR-133a and miR-19b was reduced in the myocardial and serum samples from Every bit patients compared to command subjects (Fig. i). No differences were found in serum miR-21 expression between AS patients and command subjects (data not shown), and thus no farther analysis of this microRNA was performed.
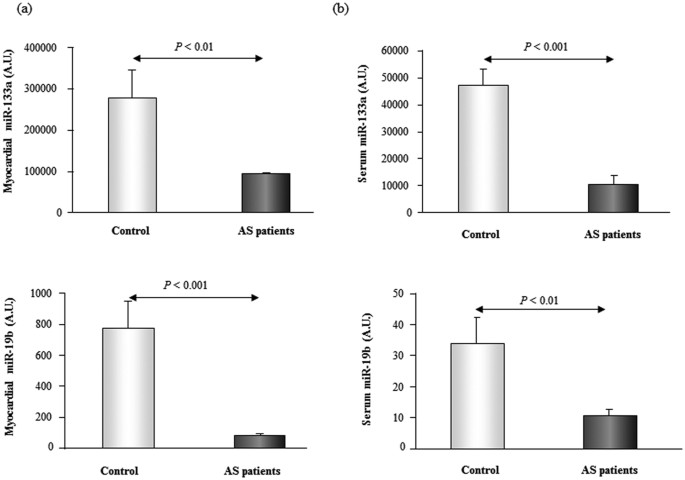
Relative expression of miR-133a and miR-19b in myocardium [Panel (a)] and serum [Console (b)] from patients with aortic stenosis (AS) and control subjects. Information are shown as mean ± SEM. A.U. ways arbitrary units.
No associations were found between myocardial and serum levels of miR-133a. In contrast, a direct correlation was observed for miR-19b between its expression in myocardial tissue and in peripheral claret in all Equally patients (r = 0.375, p < 0.05). Thus, only the associations of this microRNA with cardiac parameters were furtherly evaluated.
Associations of miR-19b with cardiac parameters
Myocardial miR-19b was inversely correlated with CVF (r = −0.409, p < 0.05), as well every bit with LOX, and CCL in As patients (Fig. 2). Moreover, myocardial miR-19b was inversely correlated with KLV in AS patients (Fig. two). Serum levels of miR-19b were too inversely correlated with LOX, CCL and ThouLV in Equally patients (Fig. 3).
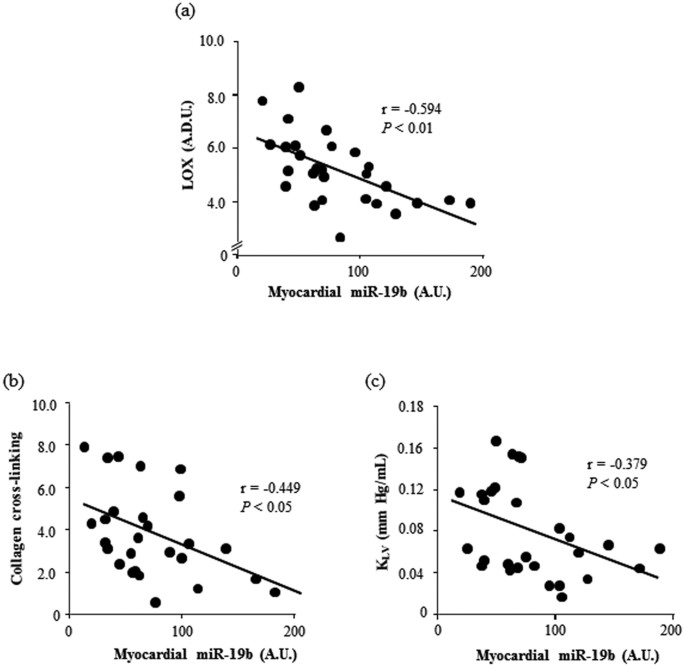
Changed correlations betwixt myocardial miR-19b expression and myocardial LOX protein [linear fit: −1.823x + 6.761; panel (a)], collagen cross-linking [linear fit: −2.224x + 5.626; panel (b)] and left ventricular chamber stiffness constant (MLV) [linear fit: −0.042x + 0.113; panel (c)] in patients with aortic stenosis. A.D.U. ways capricious densitometric units, A.U., arbitrary units. P values are for bivariate correlation assay.
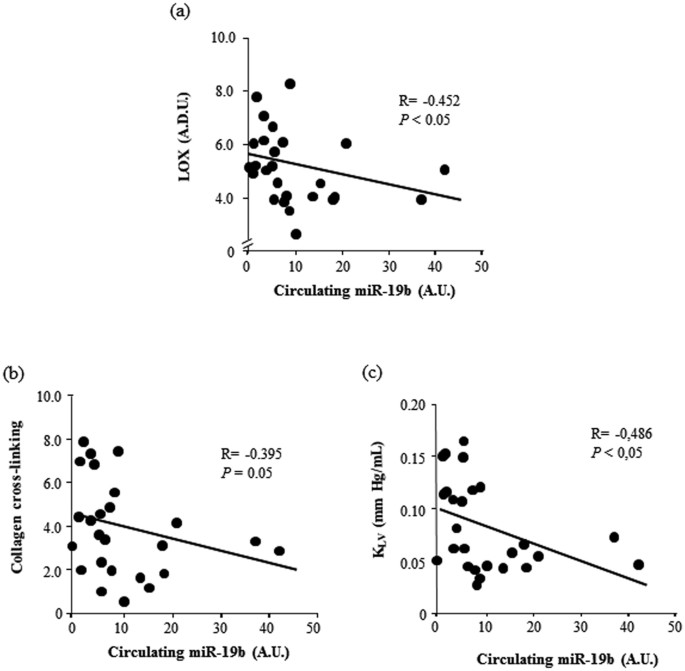
Inverse correlations between serum miR-19b expression and myocardial LOX poly peptide [linear fit: −0.037x + five.587; panel (a)], collagen cross-linking [linear fit: −0.059x + 4.505; console (b)] and left ventricular chamber stiffness constant (1000LV) [linear fit: −0.002x + 0.100; console (c)] in patients with aortic stenosis. A.D.U. means arbitrary densitometric units, A.U., arbitrary units. P values are for bivariate correlation analysis.
Interestingly, Equally patients with HF showed decreased myocardial (p < 0.01) and serum (p < 0.05) miR-19b, compared to Every bit patients without HF (Table three). Moreover, logistic regression analysis showed that decreased myocardial miR-19b was associated with the presence of HF (OR 0.949, 95% CI 0.91–0.99; p < 0.05), independently of age, gender and ejection fraction.
In vitro studies
Equally expected, miR-19b expression was decreased (p < 0.01) in HDF cells transfected with antimiR-19b (0.01 ± 0.01 A.U.) compared to those transfected with control oligonucleotides (1.06 ± 0.eighteen A.U.).
Some studies have shown that active LOX is also present in the cytosol of fibroblasts27, in fact in cell lysates we mainly detected the pro-enzyme but we could too notice the agile course. Inhibition of miR-19b significantly increased (p < 0.05) the expression of LOX protein in HDF cells (Fig. 4). Although a slight increase in LOX mRNA levels was observed in HDF cells transfected with antimiR-19b, the differences were non statistically significant (Fig. four).
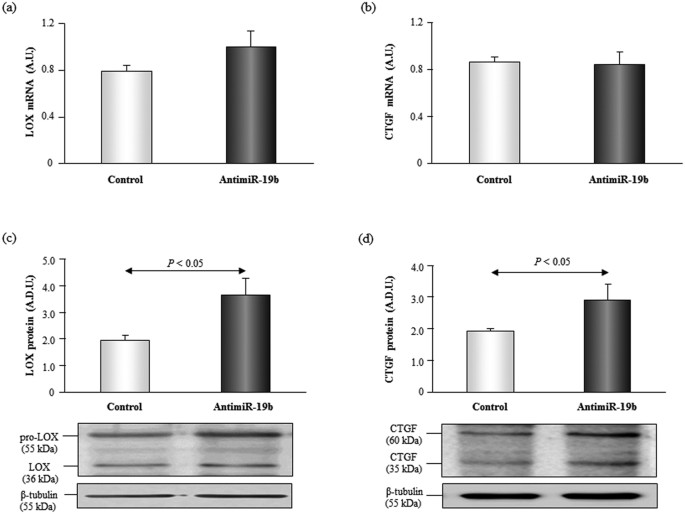
Expression of LOX mRNA and protein [panels (a) and (c)] and CTGF mRNA and poly peptide [panels (c) and (d)] in developed human dermal fibroblasts transfected with control oligonucleotides or with anti-miR-19b. Representative images of Western blots for pro-LOX (55 kDa) and LOX (36 kDa), CTGF (60 kDa and 36 kDa) and β-tubulin (55 kDa) in both groups of transfected cells. Data are shown as mean ± SEM. A.D.U. ways arbitrary densitometric units.
In silico analysis co-ordinate to the databases mentioned in the Methods section indicated that CTGF is a potential target for miR-19b, therefore, we analyzed its protein expression in HDF transfected cells. CTGF protein expression was increased (p < 0.05) in HDF cells transfected with antimiR-19b compared to command cells, whereas no changes were found in CTGF mRNA levels (Fig. iv).
Discussion
The principal findings of the present report are the following: (1) Decreased myocardial and serum miR-19b expression is present in AS patients, namely in those with HF; (2) Both myocardial and serum miR-19b are inversely associated with myocardial LOX protein and CCL, also as with LV stiffness in AS patients; (three) miR-19b inhibition increased the expression of CTGF and LOX protein in human being adult fibroblasts.
Although down-regulation of myocardial miR-19b expression has been reported previously in AS patients23, findings here reported expand this information on this microRNA showing that miR-19b was abnormally decreased not only in the myocardium but also in the blood of these patients, and namely in those with HF. In addition, we written report for the first time that decreased miR-19b was associated with increased myocardial LOX and CCL in As patients. These associations suggest that miR-19b may modulate the enzyme LOX which determines CCL14. In fact, our in vitro data suggest a role of miR-19b in the regulation of LOX expression in human fibroblasts. Although the underlying mechanisms need to be fully elucidated, we report that miR-19b inhibition also increases CTGF expression in fibroblasts. Coherently, miR-19b has been shown to regulate CTGF in ageing-associated HF both in a rodent model and in patients20. Interestingly, it has been reported that CTGF regulates LOX poly peptide expression and activeness in cardiac fibroblasts28,29. Therefore, information technology may be speculated that miR-19b might control LOX expression in human being fibroblasts via CTGF regulation.
MiR-19b belongs to the miR-17-92 cluster which includes miR-17, miR-18, miR-19a, miR-19b, miR-twenty and miR-9230. Different components of the 17–92 cluster have been reported to regulate angiogenesis31,32, cardiomyocyte proliferation33 and myocardial fibrosisxx. A recent study has shown that a decreased expression of all members of the cluster, is involved in the development of pulmonary fibrosis34. Three paralog miR-17-92 clusters take been described in humans, in chromosomes 13, X and vii35. Although some members of the cluster are located in chromosome X (eastward.grand. miR-19b-2) no differences were observed between males and females in miR-19b expression neither in myocardium nor in serum (Supplementary results).
The potential pathophysiological impact of the association betwixt miR-19b, LOX and CCL is given by our observation that these parameters were associated with LV sleeping room stiffness and that all of them were associated with the presence of HF in AS patients. In this regard, CCL has been found to be direct associated with cardiac dysfunction and LV bedchamber stiffness in hypertensive patients with HF12. Moreover, CCL was likewise associated with some hallmarks of the severity of HF (i.e. estimated pulmonary capillary wedge pressure and NT-proBNP) and with a higher risk of hospitalization for HF in patients with this syndrome12,13. Taken together, all these data back up the hypothesis that myocardial miR-19b could be involved in LV dysfunction by facilitating LOX-mediated CCL that results in the formation of stiff collagen fibers in the myocardium of patients with Every bit and HF. This is further supported by the association between myocardial miR-19b and the presence of HF observed in AS patients, independently of possible confounding factors like historic period, sex and ejection fraction.
On the other manus, the associations here reported between circulating miR-19b and myocardial parameters, namely CCL, may exist of potential clinical interest. In fact, whereas several circulating molecules accept been proposed equally biomarkers of the quantity of myocardial fibrosis (i.e. CVF)36, upwards to at present there is but one proposed biomarker of CCL (i.due east. the serum ratio of C-last telopeptide of collagen blazon I to matrix metalloproteinase-i)13. Therefore, although validation studies in larger independent cohorts are necessary, circulating miR-19b could be another biomarker of the quality of myocardial fibrosis (i.due east. CCL), at least in AS patients. In this regard, it must be noted that a direct correlation was found between myocardial and serum miR-19b suggesting the fractional cardiac origin of serum miR-19b in AS patients.
Regarding the other microRNAs found altered in this study, we confirmed that miR-133a is down-regulated in the myocardium of patients with AS37,38. However, the lack of association of this microRNA between the myocardium and the serum does non support its potential role equally biomarker of myocardial fibrosis in Equally patients. Finally, although Villar et al.xvi showed that circulating and myocardial miR-21 levels were increased in AS patients no changes in circulating miR-21 were observed in the current study, probably due to differences in the clinical characteristics of the patients (e.chiliad. differences in the frequency of patients treated with β-blockers) that might influence the expression of this microRNA39.
Study limitations
Nosotros are enlightened that this report presents some limitations. First, it involved a relatively low number of patients, but since it was a pilot study involving myocardial biopsies, the design is appropriate. 2nd, we performed biopsies of the interventricular septum to assess the effects of LV pressure level overload. Nonetheless, as previously shown40, fibrosis present in the septum from pressure overloaded man hearts is representative of fibrosis existing in the costless wall. Third, although the methodology was standardized, differences in geographical sites betwixt sample drove and processing could contribute to variability in the results. Moreover, the direct comparison of microRNA expression in myocardium and serum could be afflicted by the use of unlike types of controls for microRNA normalization: biological [U6] for tissue and technical [cel-miR-39] for the serum. Even so, these methods for normalization are unremarkably used16,26,41. Fourth, we have focused our written report in some of the chief microRNAs involved in myocardial fibrosis, due to sample limitation, notwithstanding nosotros cannot discard that other fibrosis-related microRNAs could play a role in this process37,42,43. Fifth, although previously used16, the isolation of serum microRNAs using Trizol is not the optimal method and several small RNAs may take been lost during the extraction process.
Conclusions
We report that myocardial levels of miR-19b were abnormally decreased in AS patients and were associated with an backlog of LOX and CCL, every bit well equally increased LV stiffness, namely in those patients with HF. Although descriptive in nature these findings enhance the possibility that miR-19b participates in the alterations of the myocardial collagen that impair LV mechanics and contribute to HF in Equally.
On the other hand serum miR-19b was likewise decreased in AS patients and was associated with its myocardial expression. Moreover, decreased serum miR-19b was also associated with excessive myocardial LOX and CCL, and increased LV stiffness, peculiarly in patients with HF. Thus, circulating miR-19b emerges as a potential biomarker of the alterations of myocardial collagen present in AS patients with HF.
Methods
Patients and sample acquisition
All subjects gave written informed consent to participate in the report, and the Institutional Ethics Committee of the Virgen de la Victoria University Infirmary (Málaga, Kingdom of spain) approved the protocol. The written report conformed to the principles of the Declaration of Helsinki.
The study population consisted of 28 patients with severe isolated AS referred for surgical aortic valve replacement to the Virgen de la Victoria Academy Hospital as previously described26. The diagnosis of HF was made in thirteen patients (46%) on a clinical basis, by the presence of, at least, 1 major and 2 small Framingham criteria44 and was confirmed by echocardiographic alterations in cardiac morphology and function and by the presence of elevated levels of the amino-terminal pro-encephalon natriuretic peptide (NT-proBNP)45. Patients with cardiac valve diseases other than AS, history of astute myocardial infarction, a significant stenosis (>50%) in 1 or more coronary arteries in the angiography, or disorders associated with alterations in collagen turnover were excluded after consummate medical examination. (For details encounter the Supplementary Information).
Blood and myocardial samples were obtained during surgical replacement of the aortic valve. Blood samples were obtained from the left antecubital vein at the time of valve replacement and stored at −20 °C. Two myocardial samples were obtained from the interventricular septum, one sample was immediately stock-still in 4% buffered formalin and embedded in paraffin for the determination of histomorphological parameters, and the other sample was divided into two smaller pieces and frozen separately in liquid nitrogen post-obit different processing for poly peptide or RNA isolation.
Nineteen good for you subjects were used as controls for venous blood determinations, and x necropsies from subjects who died from causes other than cardiovascular diseases were used as controls for myocardial parameters. All of these subjects were collected at the University of Navarra Clinic.
Echocardiographic assessment
Parameters assessing LV morphology and function were measured from M-manner recordings using leading edge methodology according to the American and European Societies of Echocardiography criteria. (For details run into the Supplementary Information).
NT-proBNP determination
Plasma NT-proBNP was measured by an enzyme-linked immunosorbent assay using a commercial kit (Biomedica Gruppe). All determinations were performed in indistinguishable. The inter- and intra-assay coefficients of variation were 8 and 5%, respectively.
Myocardial fibrosis assessment
CVF was determined by quantitative morphometry with an automatic prototype analysis organization (Prison cell, Soft Imaging System) in sections stained with Picrosirius Red (Sirius Reddish F3BA in aqueous picric acid), every bit previously reported40. All measurements were performed in indistinguishable past 2 contained observers. The inter- and intra-observer coefficients of variation were <4%.
To assess CCL an enzymatic and colorimetric procedure was used to evaluate insoluble (cross-linked) and soluble collagen, every bit previously described11. The CCL was calculated as the ratio between insoluble and soluble collagen. (For details see the Supplementary Information).
Protein expression of LOX was analyzed by Western absorb in myocardial samples visualized with a chemiluminescence organization (GE Healthcare) and analyzed using an automated densitometer (GS-800 Calibrated Densitometer, Bio-Rad). (For details run into the Supplementary Data).
Myocardial microRNA analysis
Myocardial RNA was isolated with TRIzol reagent (Invitrogen) according to manufacturer's recommendations (using 10 μg of glycogen to enhance the efficiency of RNA extraction) and the pellet was resuspended in forty μL of nuclease-gratis h2o. RNA concentration was determined with a NanoDrop ND-1000 Spectrophotometer. Reverse transcription (RT) of myocardial microRNAs was performed with a fixed volume of 4 μL of RNA using the TaqMan MicroRNA Reverse Transcription Kit (Applied Biosystems) and a pool of specific primers for miR-19b, miR-133a, miR-21, miR-29, miR-1, miR-208a, miR-499-5p and for snU6 (each primer diluted one:100 in the pool). The cDNA obtained was pre-amplified for 12 cycles using the TaqMan PreAmp Master Mix Kit (Applied Biosystems) and a puddle of specific TaqMan microRNA assays (each assay diluted one:100 in the pool) as previously described26. PCR of these pre-amplified microRNAs was performed with specific TaqMan microRNA assays. The fluorescent signal was detected with a 7900HT Fast Real-Time PCR Organisation (Practical Biosystems) and analyzed with the SDS 2.2.two software. The snU6 RNA was analyzed with a specific TaqMan probe every bit an endogenous control. The fluorescent signal was detected and analyzed as previously described. MicroRNA expression was calculated past the 2−ΔΔCt method relative to snU6 RNA and data were presented as arbitrary units (A.U.).
Serum microRNA analysis
Circulating microRNA levels were determined past existent-time RT-PCR. RNA was isolated from 150 μL of serum with TRIzol LS reagent (Invitrogen) according to manufacturer's recommendations (using 10 μg of glycogen to enhance the efficiency of RNA extraction). Constructed versions of the C. elegans microRNA 39 (cel-miR-39) were spiked into serum, at a abiding amount of 25 fmol, after the addition of the Trizol LS reagent to the samples. The RNA pellet was resuspended in 40 μl of nuclease-gratis h2o and RNA concentration was adamant with a NanoDrop ND-chiliad Spectrophotometer (NanoDrop Tech). RT was performed with a fixed volume of 4 μL of RNA using the TaqMan MicroRNA Contrary Transcription Kit (Applied Biosystems) as indicated for myocardial tissue. The cDNA obtained was pre-amplified as described above for myocardial tissue and amplified using the specific TaqMan microRNA assays (Life Technologies). The fluorescent signal was detected and analyzed as previously described. The Ct values from existent-time polymerase chain reaction (PCR) assays greater than 40 were treated as forty. MicroRNA expression was calculated by the 2−ΔΔCt method relative to cel-miR-39 expression and presented every bit arbitrary units (A.U.).
In silico analysis
Prediction of potential targets of the miRNA of interest was performed using the databases microRNA.org (http://www.microrna.org), miRDB (http://mirdb.org/miRDB), Targetscan vi.ii (http://www.targetscan.org), mirTarBase (http://mirtarbase.mbc.nctu.edu.tw) and TarBase (http://diana.imis.athena-innovation.gr).
In vitro studies
Adult homo dermal fibroblasts (HDF) (Thermo Fisher Scientific) were transfected with antimiR-19b or negative command oligonucleotides (16 nM) using RNAiMax Lipofectamine (Thermo Fisher Scientific) co-ordinate to manufacturer'south recommendations, and total protein and RNA were isolated after 24 hours. Intracellular protein was isolated with the M-PER mammalian protein extraction reagent (Thermo Fisher Scientific) whereas the RNA was isolated with the Maxwell® 16 LEV only RNA Purification Kit (Promega). The mRNA expression of connective tissue growth factor (CTGF) and LOX was analyzed by RT-PCR using specific Taqman assays (Thermo Fisher Scientific and Integrated DNA Technologies). Protein expression of CTGF and LOX was analyzed by Western blot. (For details come across the Supplementary Data).
Statistical assay
To analyze the differences betwixt controls and AS patients, between patients with and without HF, and betwixt two groups of cells, a Pupil's t exam for unpaired information was used once normality was demonstrated (Shapiro-Wilks examination); otherwise, a not-parametric exam (Mann-Whitney U test) was used. The association between continuously distributed variables was tested by bivariate correlation assay. Non-parametric distributed variables were examined later on logarithmic transformation. Logistic regression analysis was performed to evaluate the association betwixt quantitative and qualitative variables and the potential influence of historic period, gender and the ejection fraction was considered in the analysis. Information are expressed every bit mean ± standard error of the hateful (S.E.M.), and per centum of patients. All tests were two sided, and a value of p < 0.05 was considered statistically significant. The assay was performed using the SPSS 15.0 statistical parcel (SPSS Inc.).
Additional Information
How to cite this commodity: Beaumont, J. et al. MicroRNA-19b is a potential biomarker of increased myocardial collagen cross-linking in patients with aortic stenosis and heart failure. Sci. Rep. seven, 40696; doi: 10.1038/srep40696 (2017).
Publisher'south note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
-
16 Apr 2020
An amendment to this paper has been published and tin can be accessed via a link at the peak of the paper.
References
-
Nkomo, 5. T. et al. Burden of valvular heart diseases: a population-based report. Lancet. 368, 1005–1011 (2006).
-
Dweck, M. R., Benefaction, N. A. & Newby, D. E. Calcific aortic stenosis: a disease of the valve and the myocardium. J Am Coll Cardiol. threescore, 1854–1863 (2012).
-
Hein, S. et al. Progression from compensated hypertrophy to failure in the pressure-overloaded human heart: structural deterioration and compensatory mechanisms. Circulation. 107, 984–991 (2003).
-
Weidemann, F. et al. Touch of myocardial fibrosis in patients with symptomatic severe aortic stenosis. Circulation. 120, 577–584 (2009).
-
Krayenbuehl, H. P., Hess, O. M., Monrad, E. S., Schneider, J., Mall, K. & Turina, One thousand. Left ventricular myocardial construction in aortic valve disease before, intermediate, and tardily subsequently aortic valve replacement. Circulation. 79, 744–755 (1989).
-
Golia, Thou. et al. Influence of myocardial fibrosis on left ventricular hypertrophy in patients with symptomatic astringent aortic stenosis. Cardiology. 120, 139–145 (2011).
-
Milano, A. D. et al. Prognostic value of myocardial fibrosis in patients with severe aortic valve stenosis. J Thorac Cardiovasc Surg. 144, 830–837 (2012).
-
Azevedo, C. F. et al. Prognostic significance of myocardial fibrosis quantification past histopathology and magnetic resonance imaging in patients with severe aortic valve illness. J Am Coll Cardiol. 56, 278–287 (2010).
-
Robins, S. P. Biochemistry and functional significance of collagen cross-linking. Biochem Soc Trans. 35, 849–852 (2007).
-
Kasner, M. et al. Diastolic tissue Doppler indexes correlate with the caste of collagen expression and cantankerous-linking in heart failure and normal ejection fraction. J Am Coll Cardiol. 57, 977–985 (2011).
-
López, B. et al. Touch of treatment on myocardial lysyl oxidase expression and collagen cross-linking in patients with center failure. Hypertension. 53, 236–242 (2009).
-
López, B., Querejeta, R., González, A., Larman, Grand. & Diez, J. Collagen cantankerous-linking but not collagen amount associates with elevated filling pressures in hypertensive patients with stage C heart failure: potential office of lysyl oxidase. Hypertension. threescore, 677–683 (2012).
-
López, B. et al. Myocardial collagen cross-linking is associated with heart failure hospitalization in patients with hypertensive middle failure. J Am Coll Cardiol. 67, 251–260 (2016).
-
López, B. et al. Function of lysyl oxidase in myocardial fibrosis: from basic science to clinical aspects. Am J Physiol Centre Circ Physiol. 299, H1–H9 (2010).
-
Bauersachs, J. Regulation of myocardial fibrosis by MicroRNAs. J Cardiovasc Pharmacol. 56, 454–459 (2010).
-
Villar, A. V. et al. Myocardial and circulating levels of microRNA-21 reflect left ventricular fibrosis in aortic stenosis patients. Int J Cardiol. 167, 2875–2881 (2013).
-
Thum, T. et al. MicroRNAs in the homo heart: a inkling to fetal gene reprogramming in center failure. Circulation. 116, 258–267 (2007).
-
Matkovich, S. J. et al. Reciprocal regulation of myocardial microRNAs and messenger RNA in human cardiomyopathy and reversal of the microRNA signature by biomechanical support. Circulation. 119, 1263–1271 (2009).
-
van Rooij, Due east. et al. Dysregulation of microRNAs after myocardial infarction reveals a role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci USA 105, 13027–13032 (2008).
-
van Almen, Chiliad. C. et al. MicroRNA-18 and -nineteen regulate CTGF and TSP-1 expression in age-related heart failure. Aging Cell. 10, 769–779 (2011).
-
Karakikes, I. et al. Therapeutic cardiac-targeted delivery of miR-ane reverses pressure overload-induced cardiac hypertrophy and attenuates pathological remodeling. J Am Heart Assoc. two, e000078 (2013).
-
Matkovich, S. J. et al. MicroRNA-133a protects against myocardial fibrosis and modulates electrical repolarization without affecting hypertrophy in pressure level-overloaded developed hearts. Circ Res. 106, 166–175 (2009).
-
Ikeda, South. et al. Contradistinct microRNA expression in homo heart disease. Physiol Genomics. 31, 367–373 (2007).
-
Dawson, K. et al. MicroRNA29: a mechanistic contributor and potential biomarker in atrial fibrillation. Circulation. 127, 1466–1475 (2013).
-
Corsten, M. F. et al. Circulating microRNA-208b and microRNA-499 reflect myocardial impairment in cardiovascular disease. Circ Cardiovasc Genet. three, 499–506 (2010).
-
Beaumont, J. et al. microRNA-122 down-regulation may play a role in severe myocardial fibrosis in human aortic stenosis through TGF-beta1 up-regulation. Clin Sci. 126, 497–506 (2014).
-
Adam, O. et al. Role of miR-21 in the pathogenesis of atrial fibrosis. Bones Res Cardiol. 107, 278 (2012).
-
López, B. et al. Osteopontin-mediated myocardial fibrosis in heart failure: a role for lysyl oxidase? Cardiovasc Res. 99, 111–120 (2013).
-
Lavall, D. et al. The mineralocorticoid receptor promotes fibrotic remodeling in atrial fibrillation. J Biol Chem. 289, 6656–6668 (2014).
-
He, L. et al. A microRNA polycistron as a potential man oncogene. Nature. 435, 828–833 (2005).
-
Iaconetti, C. et al. Inhibition of miR-92a increases endothelial proliferation and migration in vitro too as reduces neointimal proliferation in vivo subsequently vascular injury. Bones Res Cardiol. 107, 296 (2012).
-
Bonauer, A. et al. MicroRNA-92a controls angiogenesis and functional recovery of ischemic tissues in mice. Science. 324, 1710–1713 (2009).
-
Chen, J. et al. mir-17-92 cluster is required for and sufficient to induce cardiomyocyte proliferation in postnatal and adult hearts. Circ Res. 112, 1557–1566 (2013).
-
Dakhlallah, D. et al. Epigenetic regulation of miR-17~92 contributes to the pathogenesis of pulmonary fibrosis. Am J Respir Crit Care Med. 187, 397–405 (2013).
-
Tanzer, A. & Stadler, P. F. Molecular evolution of a microRNA cluster. J Mol Biol. 339, 327–335 (2004).
-
López, B., González, A. & Díez, J. Circulating biomarkers of collagen metabolism in cardiac diseases. Circulation. 121, 1645–1654 (2010).
-
Duisters, R. F. et al. miR-133 and miR-xxx regulate connective tissue growth cistron: implications for a function of microRNAs in myocardial matrix remodeling. Circ Res. 104, 170–178 (2009).
-
Villar, A. Five. et al. Myocardial factor expression of microRNA-133a and myosin heavy and light chains, in conjunction with clinical parameters, predict regression of left ventricular hypertrophy after valve replacement in patients with aortic stenosis. Heart. 97, 1132–1137 (2011).
-
Hou, Y. et al. β-adrenoceptor regulates miRNA expression in rat heart. Med Sci Monit. 18, BR309-314 (2012).
-
López, B., González, A., Querejeta, R., Larman, Grand. & Diez, J. Alterations in the pattern of collagen deposition may contribute to the deterioration of systolic function in hypertensive patients with heart failure. J Am Coll Cardiol. 48, 89–96 (2006).
-
Thum, T. et al. MicroRNA-21 contributes to myocardial disease by stimulating MAP kinase signalling in fibroblasts. Nature. 456, 980–984 (2008).
-
Tijsen, A. J. et al. The microRNA-15 family inhibits the TGFβ-pathway in the heart. Cardiovasc Res. 104, 61–71 (2014).
-
Pan, Z. et al. MicroRNA-101 inhibited postinfarct cardiac fibrosis and improved left ventricular compliance via the FBJ osteosarcoma oncogene/transforming growth factor-β1 pathway. Apportionment. 126, 840–850 (2012).
-
Ho, K. K., Pinsky, J. 50., Kannel, W. B. & Levy, D. The epidemiology of heart failure: the Framingham Study. J Am Coll Cardiol. 22, 6A–13A (1993).
-
Ponikowski, P. et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Chore Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Middle Failure Association (HFA) of the ESC. Eur Middle J. 37, 2129–2200 (2016).
Acknowledgements
The authors thank Ana Igea for technical support and Maribel Zudaire for the help provided in the methodology used in microRNAs analysis. This piece of work was funded through the Ministry of Economic system and Competitiveness, Instituto de Salud Carlos III, Spain (grants RD12/0042/0009, CIBER-CV CB16/11/00483 and PI15/01909), the Castilian Order of Cardiology (research grant) and the European Matrimony (HOMAGE projection grant Health-2012-305507; FIBRO-TARGETS project grant Health-2013-602904).
Author data
Affiliations
Contributions
Javier Beaumont and Begoña López participated in the conception, design, analysis and estimation of data, drafting and final approval of the manuscript. Susana Ravassa, Nerea Hermida, Gorka San José, Idoia Gallego, Félix Valencia and Juan José Gómez-Doblas partipated in the analysis and interpretation of data, drafting and terminal approval of the manuscript. Eduardo de Teresa performed the critical revision of the manuscript for important intellectual content and final approving. Javier Díez and Arantxa González participated in the formulation, blueprint, analysis, drafting and revision of the manuscript and final approval.
Corresponding authors
Ideals declarations
Competing interests
The authors declare no competing fiscal interests.
Supplementary data
Rights and permissions
This piece of work is licensed under a Artistic Commons Attribution four.0 International License. The images or other third party material in this article are included in the commodity's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included nether the Creative Commons license, users will demand to obtain permission from the license holder to reproduce the textile. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
Reprints and Permissions
Nearly this commodity
Cite this commodity
Beaumont, J., López, B., Ravassa, S. et al. MicroRNA-19b is a potential biomarker of increased myocardial collagen cantankerous-linking in patients with aortic stenosis and middle failure. Sci Rep vii, 40696 (2017). https://doi.org/x.1038/srep40696
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/ten.1038/srep40696
Further reading
Comments
By submitting a comment yous agree to abide past our Terms and Community Guidelines. If you notice something abusive or that does not comply with our terms or guidelines please flag information technology as inappropriate.
Source: https://www.nature.com/articles/srep40696
0 Response to "Life Science 7 Student Text 4th Edition 19b Section Review"
Enviar um comentário